Atomic Orbitals
At GCSE, electrons fill shells, in the order 2,8,8,2 as GCSE only takes us as far as calcium. The sequence for AS level is very similar, the main difference is terminology. Shells are more commonly called 'energy levels'.
Energy levels are numbered 1, 2, 3 etc. which coincides with the periods of the Periodic Table. These main energy levels are split into sub-levels, s, p, d and f, (standing for sharp, principle, diffuse and fundamental).
· Energy level 1 contains an s sub-level.
· Energy level 2 contains an s sub-level and a p sub-level.
· Energy level 3 contains an s sub-level, a p sub-level and a d sub-level.
Each sub-level contains orbitals. An orbital is a region of space where an electron may be found. A maximum of 2 electrons can fit in an orbital.
· s sub-levels contain only one orbital.
· p sub-levels contain 3 orbitals (called px, py and pz).
· d sub-levels contain 5 orbitals (called dxy, dxz, dyz, dz2 and dx2-y2).
Energy levels are numbered 1, 2, 3 etc. which coincides with the periods of the Periodic Table. These main energy levels are split into sub-levels, s, p, d and f, (standing for sharp, principle, diffuse and fundamental).
· Energy level 1 contains an s sub-level.
· Energy level 2 contains an s sub-level and a p sub-level.
· Energy level 3 contains an s sub-level, a p sub-level and a d sub-level.
Each sub-level contains orbitals. An orbital is a region of space where an electron may be found. A maximum of 2 electrons can fit in an orbital.
· s sub-levels contain only one orbital.
· p sub-levels contain 3 orbitals (called px, py and pz).
· d sub-levels contain 5 orbitals (called dxy, dxz, dyz, dz2 and dx2-y2).
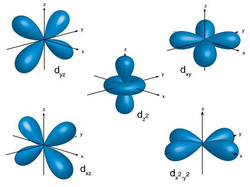
Image via de.wikipThe d sub-shell
There are five d orbitals, which are called dxy, dyz, dxz, dx2-y2 and dz2.
Four of them look like four leaf clover. One looks like a dumbbell in a doughnut.
You don't need to know these for AS Chemistry but they're quite interesting!
Image via de.wikipedia.org
Electronic Structures of 3 common elements
There are five d orbitals, which are called dxy, dyz, dxz, dx2-y2 and dz2.
Four of them look like four leaf clover. One looks like a dumbbell in a doughnut.
You don't need to know these for AS Chemistry but they're quite interesting!
Image via de.wikipedia.org
Electronic Structures of 3 common elements
- At GCSE, you would have written the electronic configuration of lithium (Li) as 2,1 because it has 3 electrons - 2 in the first shell and 1 in the outer shell.
- At AS, we now write the electronic configuration of lithium as 1s2 2s1 which means that there are two electrons in the 1s subshell and one electron in the 2s subshell.
- At GCSE, you would have written the electronic configuration of oxygen (O) as 2,6 because it has 8 electrons - 2 in the first shell and 6 in the outer shell.
- At AS, we now write the electronic configuration of oxygen as 1s2 2s2 2p4 which means that there are two electrons in the 1s subshell, two electrons in the 2s subshell and four electrons in the 2p subshell.
- As GCSE you would have written the electronic configuration of aluminium (Al) as 2,8,3 because it has 13 electrons - 2 in the fist shell, 8 in the second shell and 3 in the outer shell.
- At AS, we now write the electronic configuration of Aluminium (Al) as 1s2 2s2 2p6 3s2 3p1 which means that there are two electrons in the 1s subshell, two electrons in the 2s subshell, six electrons in the 2p subshell, two electrons in the 3s subshell and one electron in the 3p subshell.